The Role of Anterior Segment Imaging in I. Glaucoma Diagnosis

Helena Filipe, MD, MSc
Summary
Ultrasound biomicroscopy (UBM) and anterior segment optical coherence tomography (AS-OCT) provide comprehensive information on the structural aspects of the anterior segment. This paper aims to appraise the role of anterior segment imaging in the diagnosis of glaucoma.
The illustrative images were acquired utilizing both technologies.
We herein (I) Present the morphology and topography of the anterior segment structures of eyes with angle closure disease as shown by both technologies and explain how this understanding discloses pathophysiology aspects leading to effective management; (II) Appreciate the effects of light, accommodation and age on the ACA; (III) Describe the relevance of the structural aspects in effectively managing pigmentary glaucoma. (IV) Describe several biometric descriptors to screen and follow up glaucoma.
Introduction
Optical biomicroscopy and gonioscopy have been classically used to assess the anterior segment and ACA structures and how they relate.1
The innovative development of anterior segment imaging has allowed visualizing the anterior segment structures and aspects relevant to the diagnosis and management of glaucoma, which could not be conveyed by conventional techniques.2
The physical rationale for AS-OCT is based on low-coherence interferometry. It is based on the emission of an infrared light beam by a super luminescent diode, which is split in two: one sub-beam is directed towards the biological sample and the other one to a reference mirror. The analysis of the delay of these two sub-beams reflection by these structures (time-domain technology or TD-OCT) or the spectral analysis of the reflected interferometry signal (spectral Fourier-domain technology or SD/FD-OCT) enables the reconstitution of high-resolution bidimensional cross sectional images. The former is used by the OCT Visante® (Carl Zeiss Meditec, Dublin, CA, USA), whereas the latter is used by SD/FD-OCT (Heidelberg Engineering, Heidelberg, Germany). The swept-source OCT or SS-OCT is based on the SD-OCT and allows a full 360º assessment of the ACA.1,3,5
The ultrasound biomicroscopy or UBM is based on the emission of a high-frequency ultrasound beam (35, 50 and 80 MHz) with low tissue penetration. The reflection of the ultrasound beam at tissue interfaces of the anterior segment structures produces echoes and allows the acquisition of high-resolution cross sectional images. The technique requires the ultrasound transducer to be immersed in a saline water bath filling either an eyecup or a ClearScan® probe cover applied onto the eye.
The ultrasound waves cross well the pigmented epithelia, thus offering high-resolution images of structures behind the iris, as the ciliary body is. The same does not apply to light beams, which are highly absorbed by the posterior pigment layer of the iris.1,4,5,6. Table 1 summarizes the overlapping performance attributes of both technologies and highlights the advantages and pitfalls of each of them.
Table 1 Clinical applications, advantages and disadvantages of UBM and AS-OCT.
|
UBM and AS-OCT |
UBM |
AS- OCT |
|---|---|---|
|
Performance |
Advantages |
|
|
Provide evidence on contributing mechanisms and principal structures causing angle-closure glaucoma to decide the best effective treatment
Characterize quantitatively anterior segment using measurement parameters as valuable screening, diagnostic and treatment assessment tools
Explain how the ciliary body/iris complex - ACA amplitude responds to the effect of age, accommodative and lighting stimuli
Assess glaucoma surgery outcomes |
Provides high-resolution cross sectional images of anterior segment and ACA, including structures behind the iris such as the ciliary body, zonule, pars plana, anterior lens face, lens equator and anterior vitreous
Enables visualization of morphology and topography changes of ACA structures associated with lighting conditions, drugs, diagnostic indentation and age |
Provides high-resolution cross sectional images of anterior segment to the iris-lens diaphragm
Is easy to operate
Does not require much collaboration
Offers fast acquisition
Provides exact image location
Can be used in recent post-operative period and ocular trauma |
|
Disadvantages |
||
|
Requires Supine position Immersion technique Using the ClearScan® filled with saline adapted to the probe may facilitate the former. May cause Discomfort Abrasion and infection Inadvertent indentation Can be time-consuming Demands a Trained operator Collaborating patient |
Does not provide much detail of the structures posterior the iris pigment epithelium
Allows a panoramic view of the anterior segment, to date only the OCT Visante®
Offers either high-resolution images of the cornea or the ACA generally in different frames using distinct acquisition modes with technologies other than the Visante®
|
|
Clinical Applications of Anterior Segment Imaging in Glaucoma
I. Morphology and Topography of the structures of of the anterior segment in eyes with Angle Closure Disease
A. Angle Closure and Primary Angle Closure Glaucoma
Primary angle closure glaucoma is a heterogeneous group of eye diseases caused by a variety of mechanisms. It can be categorized according to the mainly involved anterior segment structure showing abnormalities related to size or position or as a result of abnormal forces in the posterior segment.7,8 (Table 2)
Table 2 Classification of angle closure glaucoma according to the four anatomic sites where aqueous humor flow obstruction can mainly occur
|
Site of aqueous humor obstruction |
Angle closure/glaucoma |
|---|---|
|
Iris |
Primary Pupil Block |
|
Ciliary Body |
Plateau Iris |
|
Lens |
Phacomorphic Glaucoma |
|
Suprachoroidal Fluid |
Uveal Effusion/Malignant Glaucoma |
This invariably leads to iris apposition to the trabecular meshwork and subsequent increase in intraocular pressure. High-resolution images provided by AS-OCT and especially UBM can unveil the structure preferably involved in the underlying mechanism and guide to the most effective management approach.6,9
A1 Primary Pupil Block
Aqueous humor flow resistance at the irido-lenticular contact increases the pressure in the posterior chamber, widens its area and produces an anterior iris bowing leading to angle closure.6 (Fig.1)
UBM and AS-OCT confirm diagnosis and document pressure balance restoration between both anterior and posterior chambers after laser peripheral iridotomy (LPI). Anterior segment imaging can also explain the reasons whereby this procedure may show unsuccessful in some angle closure situations such as incomplete LPI, goniosynechiae and plateau iris syndrome.
Iris-trabecular apposition disclosed by gonioscopy, UBM or AS-OCT do not necessarily imply an intraocular pressure increase.10 Contact may be intermittent or relatively short (circularly lower than 360º and longitudinally not reaching the Schwalbe's line) and the uveoscleral route may represent approximately 10% of drainage. Usually, mydriasis is associated with a decrease of the iris root aqueous volume. In eyes prone to angle-closure glaucoma, this phenomenon can be compromised and even be a precipitating factor.6 From the latter two aspects may be inferred: the relevance of assessing iris thickness and standardizing test lighting conditions when using UBM or AS-OCT for quantitative assessments.11
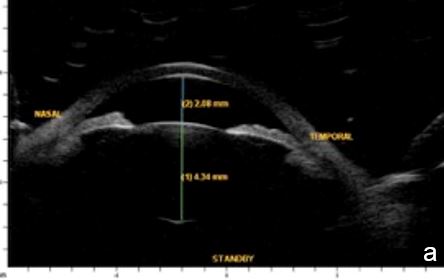
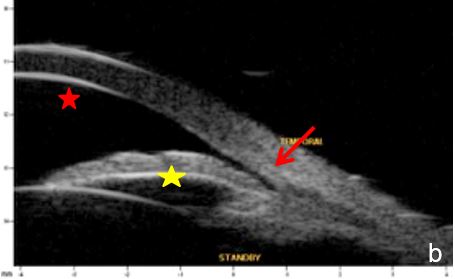
Figure 1 Primary pupil block (scleral spur= red arrow, anterior chamber=red star; posterior chamber=yellow star). a. horizontal axial section, b. radial section UBM
A2 Plateau Iris
UBM and AS-OCT show a generally thick, flat, slightly anterior bowing iris which abruptly ascends after its insertion on the ciliary body’s anterior surface assuming a "square root" appearance. Indentation gonioscopy or UBM may reveal the double-hump sign. The most peripheral hump drapes over the rigid, anteriorly positioned ciliary body, and the central rests over a long extension on the anterior lens surface. A modified dynamic gonioscopy procedure using AS-OCT allowed the assumption that ACA’s amplitude was significantly higher after indentation and that indirect signs were reliable to suggest a plateau iris.1,2 Anterior rotation of the ciliary body, associated with anterior positioning of the iris dilator’s anchorage point determines: irido-ciliary sulcus narrowing, iris apposition to the trabecular meshwork and angle closure.8 The farther away the iris dilator "joint" from the scleral spur, the lower the probability of angle closure.8 The central depth of the anterior chamber is generally normal.9 (Figs. 2 and 3)
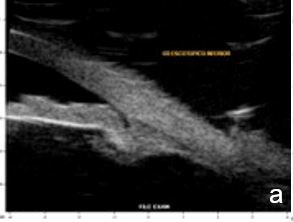
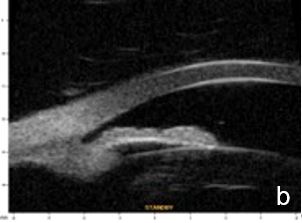
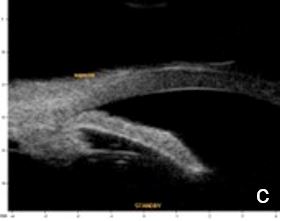
Figure 2 Plateau iris: iris root insertion on the anterior surface of the ciliary body a: anterior or internal; b: medial; c: posterior or external (UBM)
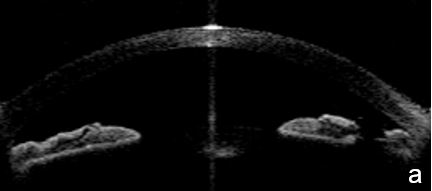
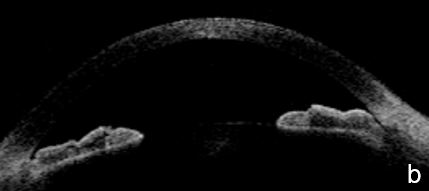
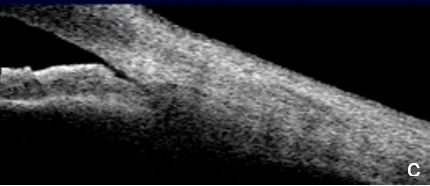
Figure 3 Plateau iris. a. Visualizing the ciliary body and how it relates with the ACA’s structures is more difficult in AS-OCT images. a. Despite LPI patency, there is still a potentially closing angle; b. anterior chamber depth within normal, flat iris with extensive, close contact to the anterior lens surface at the pupil edge. c. Steep angulation of iris root. AS-OCT
UBM and AS-OCT document argon laser peripheral iridoplasty effect on ACA opening and enable objective anterior segment measurements (Fig. 4).
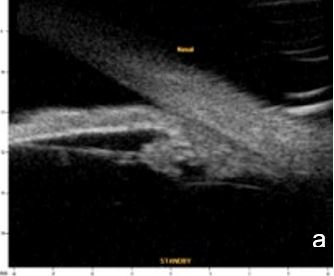
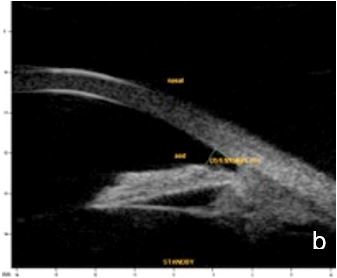
Figure 4 Effect of iridoplasty on ACA amplitude in plateau iris. a. Pre-laser b. Post-laser. AOD (500): 0.500 mm. UBM
A3 Phacomorphic Mechanism
UBM and AS-OCT can confirm the presence of an intumescent lens pushing forward the iris and ciliary body to cause iris-trabecular meshwork apposition. An increased lens vault or crystalline lens rise (CLR) elevates the probability of developing angle-closure glaucoma, regardless of lens thickness and location.13 (Figs. 5 and 6)
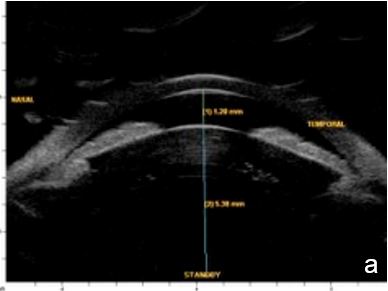
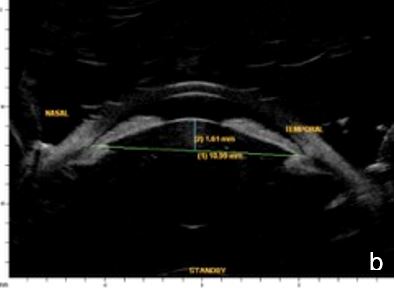
Figure 5 Phacomorphic glaucoma. a. Shallow anterior chamber (1.28 mm), high anterior-posterior lens diameter (5.38 mm), “Mount Vesuvio” iris. b. The longer line corresponds to the anterior chamber width (ACW) -10.99 mm; the shorter line corresponding to the distance from the ACW to the lens anterior pole is the lens vault or crystalline lens rise (CLR) (1.61 mm). UBM
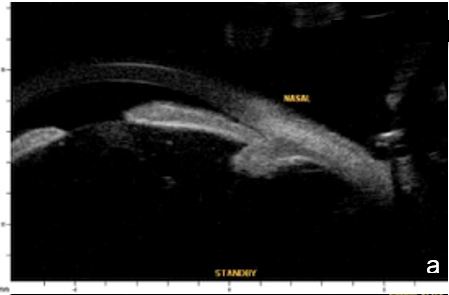
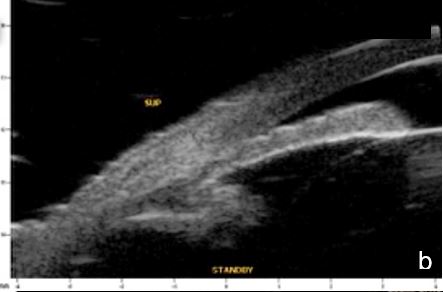
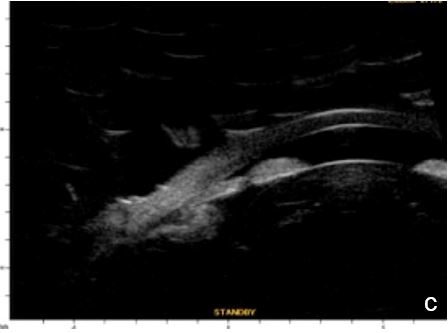
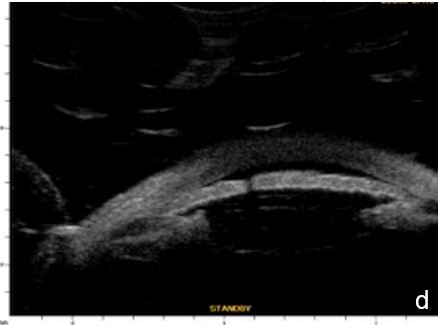
Figure 6 Phacomorphic glaucoma. a, b. radial sections showing a. ACA occludability b. angle closure, shallow posterior chamber, patent irido-ciliary sulcus and iris-lens diaphragm anteriorly positioned . c. (radial section) d. (horizontal isection) the presence of a patent IPL does not change ACA structures’ topography, which remains closed. UBM
A4 Malignant glaucoma
Some eyes from which nanophthalmos is an extreme example, are prone to angle closure due to their dimensions, to unsuccessful regulation mechanisms and vitreous resistance to fluid flow. Quigley claims that the latter determines the expansion of choroidal volume and explains why posterior lenticular pressure promotes extreme forward positioning of anterior segment structures. The mechanism afore explained leads to maximal resistance to aqueous humor flow and consequently to an intraocular pressure rise.9 UBM and AS-OCT document lens, iris and vitreous anteriorly positioned, absence of posterior chamber, markedly reduced anterior chamber depth until athalamia, and uveal effusion. (Figs. 7, 8 and 9)
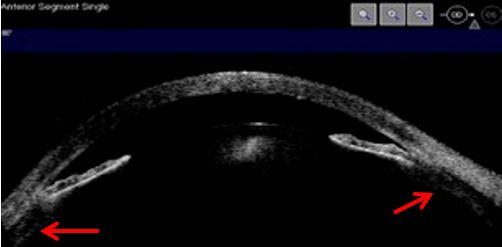
Figure 7 Corneal edema, highly shallow anterior chamber, anterior positioning of the iris-lens diaphragm, iris thinning, uveal effusion (red arrows). AS-OCT
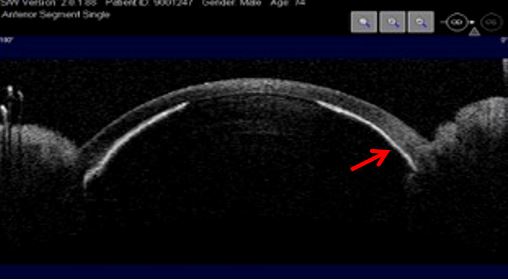
Figure 8 Extreme forward positioning of the anterior segment structures. Athalamia, iris (red arrow) and lens in direct contact with the corneal epithelium. AS-OCT
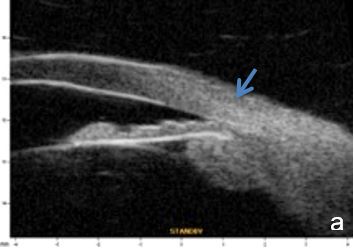
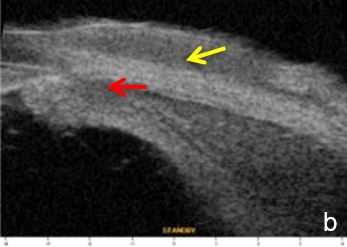
A5. Other causes of angle closure
Iris or ciliary body primary or secondary cystic or solid lesions, goniosynechia, cyclitic membrane, vitreous hernia or retinal surgery tamponade material may determine partial or intermittent angle closure that can be documented by UBM and AS-OCT. (Fig. 10)
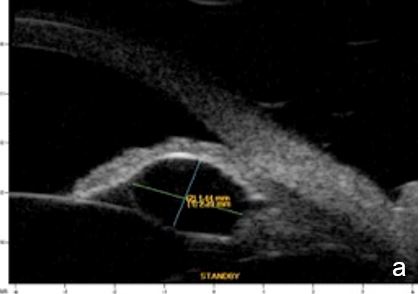
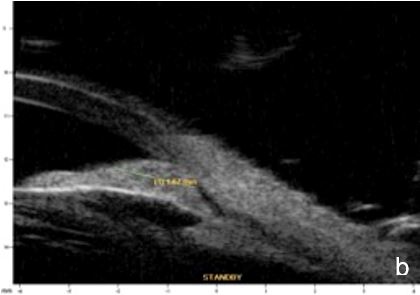
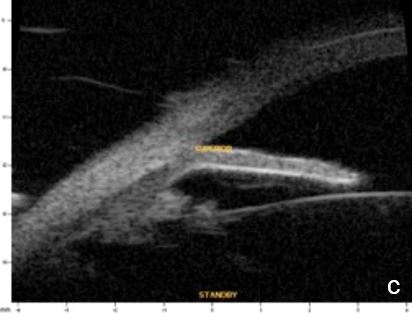
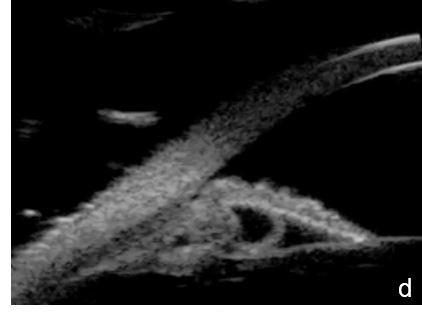
Figure 10 a. Ciliary body cystic lesion b. Iris solid lesion c. Goniosynechia d. “Pseudo plateau”. UBM
II. The effects of light, accommodation and age on the ACA
Lighting conditions may cause relevant topography variation of anterior segment structures. Hence, we usually perform the examination under scotopic/mesopic conditions. For research purposes lightening and accommodation conditions can be standardized to obtain reliable and replicable results.14 (Fig. 11)
Anterior movement of the lens was estimated to be approximately 20µm/year with increasing age. This is associated with a progressing increase of CLR, a reduced anterior chamber depth and an ACA narrowing,15 which will be clinically more relevant the higher the anatomical predisposition to angle closure, such as in hyperopia, nanophthalmos, iris plateau, etc.
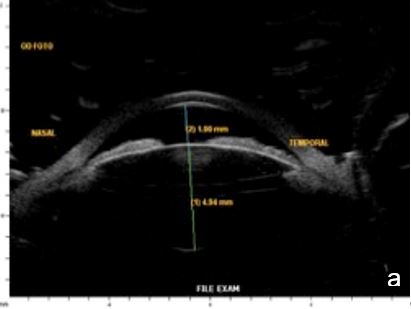
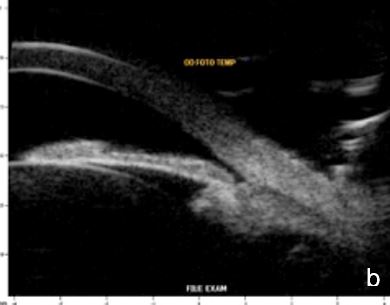
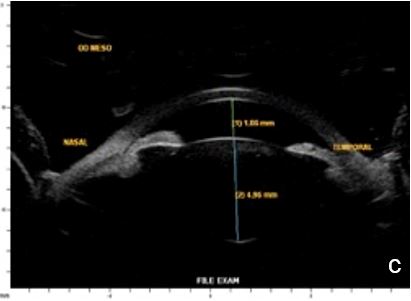
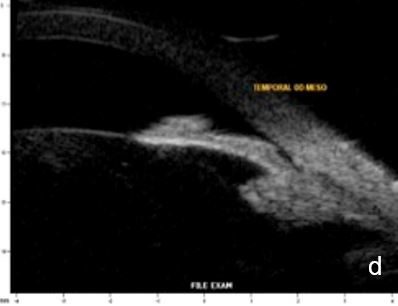
Figure 11 Effect of lighting conditions. In photopic conditons: a. axial horizontal section, b. radial section – temporal quadrant; In mesopic conditions: c. axial horizontal section, d. radial section – temporal quadrant. UBM
III. Structural aspects in Pigmentary Syndrome/Glaucoma
UBM and AS-OCT can be useful in secondary open angle glaucoma as pigmentary glaucoma and pseudoexfoliation glaucoma. The diagnostic triad of pigmentary glaucoma: Krukenberg spindle, radial transillumination defects and trabecular meshwork pigment are caused by pigment dispersion due to mechanical friction of the posterior iris surface against the zonule. Anterior segment imaging may reveal a back bowing iris touching zonular bundles, a "reverse pupillary block" and document iris flattening after LPI, confirming a pressure gradient rebalance between anterior and posterior chambers16 (Fig. 12). In pseudoexfoliation syndrome/glaucoma, UBM or AS-OCT may reveal pseudoexfoliation flecks over da lens and pupillary margin and eventually zonule fiber loss or frailty.
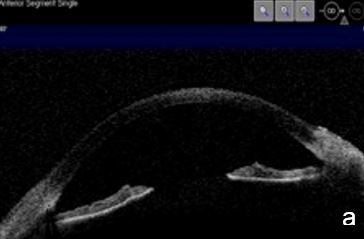
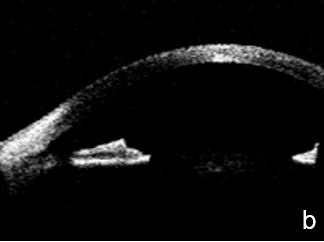
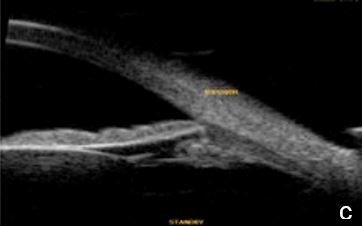
Figure 12. Pigmentary glaucoma associated with reverse pupil block. a. Back bowing iris b. Post-LPI flattened iris. AS-OCT. c. Reverse pupil block. Of note, are the visible structures behind the iris presented by UBM and not as such by AS-OCT
IV Biometric descriptors to screen and follow up glaucoma
Pavlin and associates devised the first biometric descriptors to use on UBM images. These increased objectivity, accuracy and reproducibility of anterior segment measurements.7 (Fig. 13) The concept rapidly extended to AS-OCT.1,6 (Fig. 15) The scleral spur is the landmark from which many measurement parameters are estimated. After the manual localization of the sceral spur and by applying a semi-automated software system, the ACA can be quantitatively characterized in a fast and reproducible manner6. Identifying the scleral spur in angle closure cases may be difficult, especially in the upper and lower quadrants.5,12
Research is ongoing to find one or a set of effective area or linear biometric descriptors to accurately diagnose an occludable angle and predict angle closure glaucoma.17,18
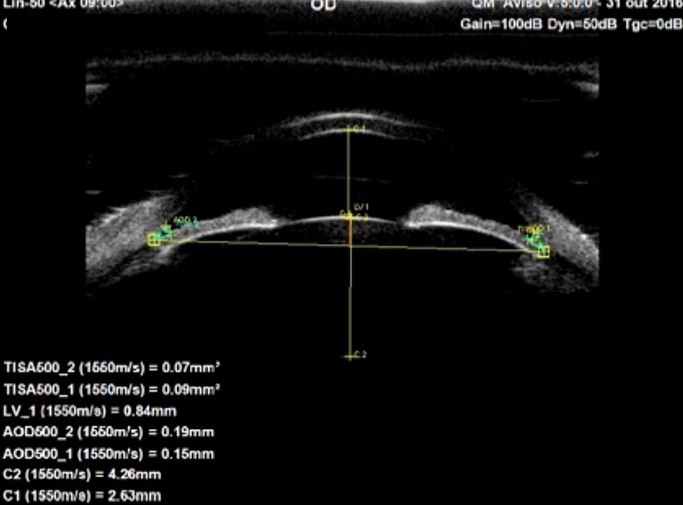
Figure 13. Biometric descriptors obtained at 0.500mm (AOD500 and TISA500) distance from the scleral spur, in which AOD: Angle Opening Distance; TISA: Trabecular Iris Surface Area; C1: Anterior Chamber Depth, Lens vault: LV1, Lens Thickness: C2. UBM (axial horizontal section)
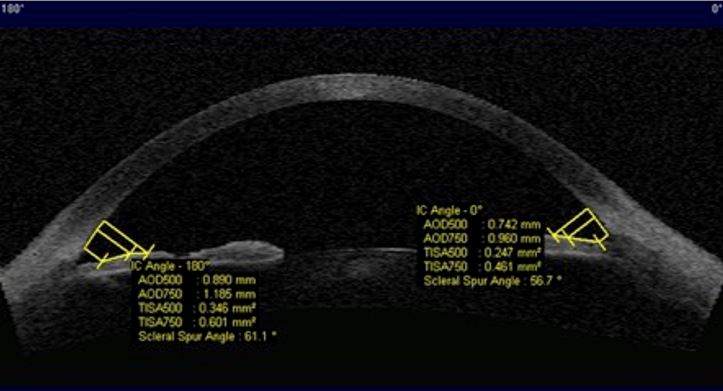
Figure 14. Biometric descriptors obtained at 0.500mm (AOD500 and TISA500) and 0.750 mm (AOD750 and TISA750) distances from the scleral spur in which AOD: angle opening distance; TISA: trabecular iris surface area. AS-OCT(axial horizontal section)
Conclusion
AS-OCT and UBM provide high-resolution cross sectional images of the anterior segment structures allowing a detailed visualization of the ACA. Both had shown to be useful in the diagnosis of glaucoma. Visualizing retroiridian structures is more challenging using AS-OCT than with UBM since the former relies on low-coherence interferometry and the pigmented posterior layer of the iris absorbs the light.
Anterior segment imaging facilitates the understanding of the underlying mechanism of the heterogenous group of angle closure glaucomas and how the effect of light, accommodation and age can influence the topography of anterior segment structures. We conclude by pointing out the relevance of quantitative anterior segment imaging.18,19,20
Acknowledgements to
Dra. Manuela Carvalho, for her insurmountable encouragement and friendship,
All Colleagues and Friends for having entrusted their Patients, whose corresponding images illustrate this paper.
----This article is being updated ----
